Recommendations below are intended as a general guideline for management of GN-BSI to provide favorable clinical outcomes while minimizing unintended consequences. Patient-specific factors should influence decisions on agent, duration, and transition to oral therapy.
This guideline does NOT apply to immunocompromised hosts (eg, Solid organ recipients; hematopoietic cell transplant recipients; patients actively receiving chemotherapy; expected prolonged neutropenia with ANC <500 cells/mL; recent CD4 count <200 cells/mL; chronic high dose corticosteroids (prednisone ≥20 mg/day or equivalent) and/or immunomodulatory therapy).
Management of GN-BSI Guidance Document
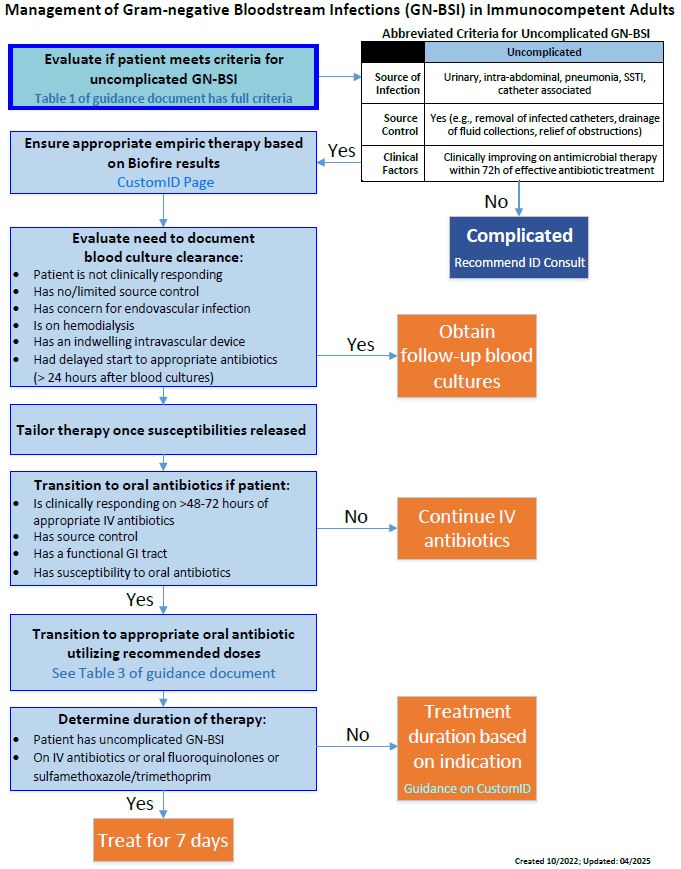
Step 1: Evaluate clinical factors indicating complicated versus uncomplicated GN-BSI (Table 1). Determine need for source control intervention and/or ID consult.
Table 1. Criteria for Uncomplicated vs Complicated GN-BSI:
|
|
Uncomplicated |
Complicated (Recommend ID Consult) |
|
Source of Infection |
Urinary tract infection (without prostatitis or renal abscess), intra-abdominal or biliary infection, pneumonia (without empyema/abscess or structural lung disease such as cystic fibrosis), SSTI, catheter-associated |
Involvement of CNS, bone or joint infection, endocarditis/endovascular infection, implanted material (e.g. hardware, devices, grafts), liver abscess |
|
Source Control |
Yes (e.g., removal of infected catheters, near complete drainage of infected fluid collections, relief of urinary or biliary tract obstructions) |
Source control not achieved |
|
Clinical Factors |
Clinically improving on current antimicrobial therapy within 72h of effective antibiotic treatment defined by:
|
Defervescence or hemodynamic stability not achieved after 72 hours.
|
SSTI: Skin and soft tissue infection, CNS: central nervous system
Note: Pathogens with limited treatment options (e.g. Pseudomonas aeruginosa, Acinetobacter spp., Stenotrophomonas maltophilia) including those with multidrug resistance may warrant ID consultation for treatment choice and duration decisions.
Step 2: Select empiric antibiotic(s) and (if indicated) document blood culture clearance
- Empiric Therapy
- Individual patient factors including severity of presentation (e.g., septic shock), previous antibiotic exposure and previous culture data should be reviewed and utilized to guide empiric therapy
- In patients with suspected intra-abdominal source or mixed soft tissue infection, metronidazole should be added to ceftriaxone, cefepime, aztreonam or ciprofloxacin for anaerobic coverage. Piperacillin/tazobactam provides adequate coverage for obligate anaerobes as monotherapy.
- Empiric antibiotic therapy recommendations based on initial speciation from rapid diagnostics can be found on Custom ID Rapid Diagnostics page.
- Clinical pharmacists may dose adjust (increase or decrease) antibiotics based on patient renal function. Full policy guidance and renal dose adjustments are located on CustomID.
- Repeat Blood Cultures: Repeat blood cultures to document clearance ONLY if at least ONE of the following are true:
- Complicated GN-BSI (See Table 1)
- Patients do NOT have appropriate clinical response within 72 hours of starting antibiotics (afebrile, hemodynamically stable, resolving leukocytosis)
- Endovascular infection or endocarditis
- Limited or no source control
- Hemodialysis
- Indwelling intravascular device (including cardiac devices)
- Delayed (>24 hours from when initial blood culture drawn) appropriate antibiotic therapy
Step 3: Tailor antibiotic therapy and/or switch to appropriately dosed oral agent based on susceptibility data and clinical response.
- Targeted Therapy: Adjust antibiotics based on antimicrobial susceptibility results to narrow spectrum agent (Table 2) UNLESS pathogen is known to have drug-resistance:
- Extended-spectrum Beta-lactamase (ESBL)-producing: Enterobacterales that are intermediate or resistant to ceftriaxone are considered ESBL-producing and should be treated with a carbapenem (meropenem 500 mg IV q6h (0.5 hr) or ertapenem 1 g IV once daily). ID approval is required.
- AmpC-producing: Patients with uncomplicated GN-BSI from ampC pathogens Enterobacter cloacae, Klebsiella (Enterobacter) aerogenes, or Citrobacter freundii can be treated with cefepime when cefepime susceptible and not cefepime resistant or susceptible dose-dependent. Due to the risk of inducible beta-lactamase production, ceftriaxone and piperacillin-tazobactam are NOT recommended for GN-BSI, even if reported as susceptible.
Table 2: De-escalating Antibiotic Therapy
|
IV (preference for narrowest-spectrum susceptible antibiotic) |
|
Ampicillin 2g IV q4h^ Ampicillin/sulbactam 3g IV q6h Cefazolin 2g IV q8h^ Ceftriaxone 2g IV q24h^ |
*See DUH CustomID for renal dose adjustments
^Addition of metronidazole required for anaerobic coverage
- Transition to Oral Therapy Considerations:
- Source control achieved
- Patient clinically improved on effective intravenous antibiotics within 48-72h (e.g., afebrile, leukocytosis improving)
- Patient has an intact and functional gastrointestinal tract
- Culture data demonstrates susceptibility to an appropriately dosed oral antibiotic (Table 3)
- Patients with GN-BSI due to ESBL- or AmpC-producing pathogens can be transitioned to oral fluoroquinolones or trimethoprim/sulfamethoxazole if they fulfill all other criteria above.
NOTE: The following oral agents should NOT be used for GN-BSI:
- Cefdinir due to unfavorable pharmacokinetics and association with worse clinical outcomes.
- Agents with limited systemic absorption or low serum levels (eg, fosfomycin, nitrofurantoin, doxycycline).
Table 3: Suggested Oral Antibiotic Therapy (consult pharmacy for patient specific dosing recommendations and for patients with obesity)
|
Agent1, 2 |
Suggested Dosing3 |
| Amoxicillin | 1000 mg PO TID |
| Amoxicillin-clavulanic acid | 875-1000 mg PO TID |
| Cephalexin^ | 1000 mg PO QID |
|
Ciprofloxacin |
500-750 mg PO BID NOTE: 750mg dose recommended for Pseudomonas spp BSIs |
|
Levofloxacin |
750 mg PO daily |
|
Trimethoprim/sulfamethoxazole |
5 mg/kg PO BID (eg, ~2 DS tablets q12h for a 70 kg patient) |
1Patient- and pathogen-specific factors should be considered when selecting an oral transition agent for gram-negative bacteremia
2Alternative oral beta-lactam agents (i.e. cefuroxime 1g q12h, cefpodoxime 400mg q12h, and cefadroxil 1g q12h) are less preferred based on agent bioavailability, ability to achieve adequate serum concentrations, and/or limited clinical data. Use may be considered on a case-by-case basis in discussion with ID/ASET.
3Doses assume normal renal function. See DUH CustomID for renal dose adjustments.
^Cefazolin susceptibility reported on urine culture does NOT predict susceptibility for GN-BSI due to use of different breakpoints. Use the cefazolin susceptibility from blood cultures to determine if oral cephalexin is active.
Step 4: Determine appropriate duration of therapy based on antibiotic agent, infection source, and route.
- Duration of Therapy: 7 days of active therapy for uncomplicated GN-BSI
- Uncomplicated GN-BSI should be treated with a 7-day total course of effective IV and/or oral therapy
- For uncomplicated GN-BSI, day 1 is the first day of therapy on an antibiotic to which the pathogen was susceptible. For patients requiring source control interventions, day 1 is the day of source control or first day of effective therapy, whichever came last.
- For patients with complicated GN-BSI, longer durations may be warranted, in agreement with existing indication-specific guidelines, documented blood clearance, and/or ID recommendation.
- In patients requiring outpatient parenteral antibiotic therapy, please consult ID to determine treatment plan prior to the day of discharge.